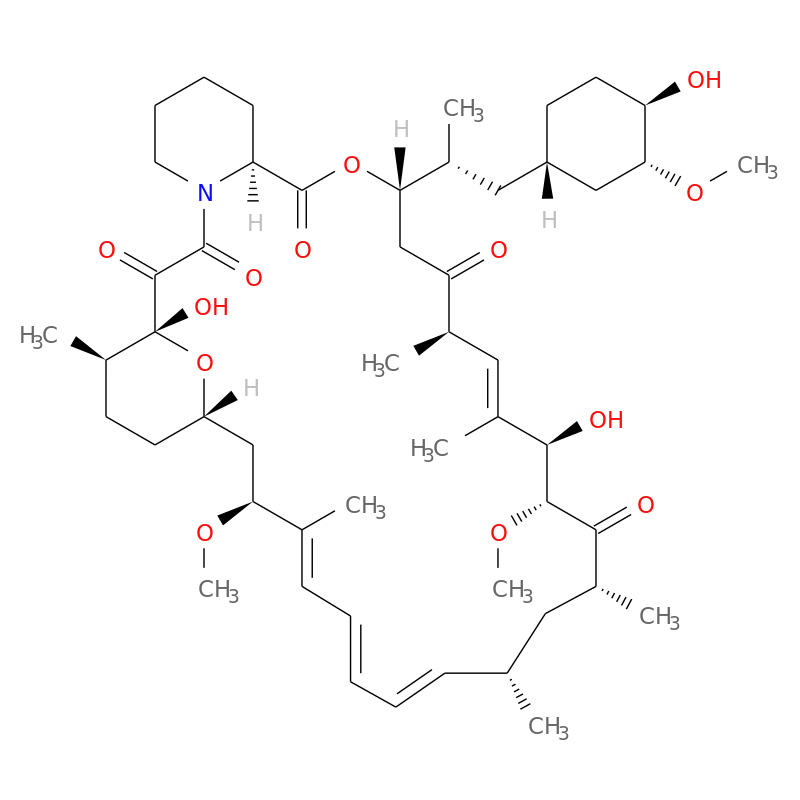
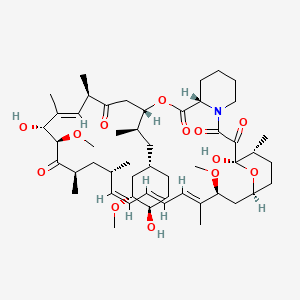
PDF) Topical sirolimus 0.1% for treating cutaneous microcystic lymphatic malformations in children and adults (TOPICAL): Protocol for a multicenter phase 2, within-person, randomized, double-blind, vehicle-controlled clinical trial
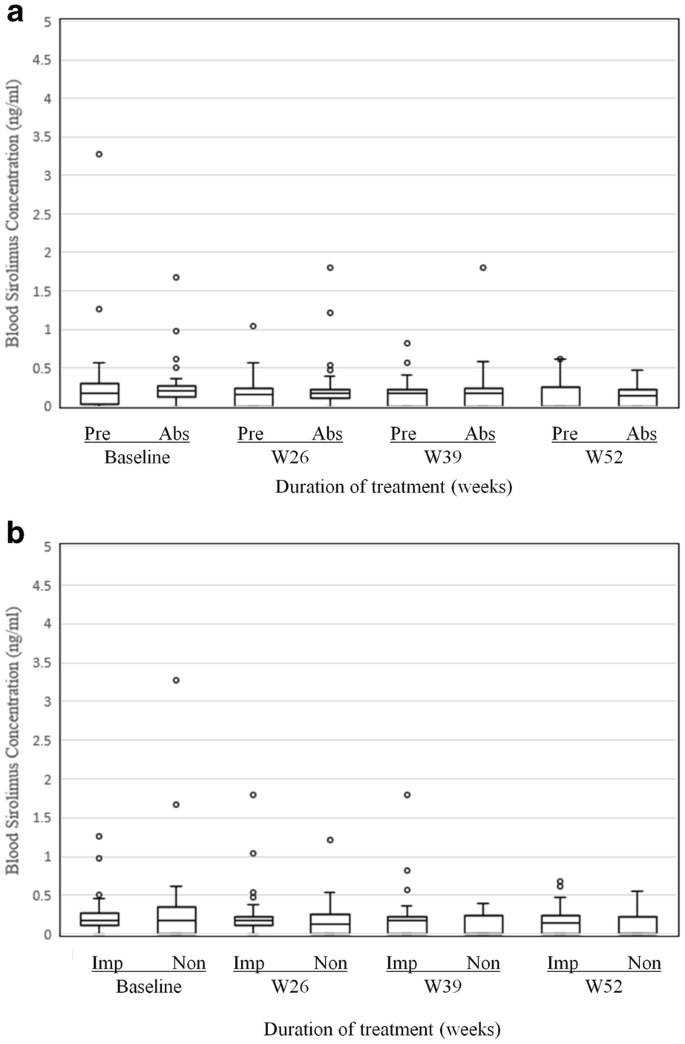
Safety and Efficacy of the Sirolimus Gel for TSC Patients With Facial Skin Lesions in a Long-Term, Open-Label, Extension, Uncontrolled Clinical Trial | Dermatology and Therapy

Baseline characteristics of 33 patients receiving sirolimus gel for... | Download Scientific Diagram

Topical rapamycin treatment alleviates the rosacea-associated symptoms... | Download Scientific Diagram

Topical sirolimus 0.1% for treating cutaneous microcystic lymphatic malformations in children and adults (TOPICAL): protocol for a multicenter phase 2, within-person, randomized, double-blind, vehicle-controlled clinical trial | Trials | Full Text

A case of microcystic lymphatic malformation successfully treated with topical sirolimus - Çalışkan - 2018 - Dermatologic Therapy - Wiley Online Library

Polymeric micelle formulations for the cutaneous delivery of sirolimus: A new approach for the treatment of facial angiofibromas in tuberous sclerosis complex - ScienceDirect

Long‐term treatment of cutaneous manifestations of tuberous sclerosis complex with topical 1% sirolimus cream: A prospective study of 25 patients | Semantic Scholar

Rapamycin Skin Cream - US Pharmacy that is Selling it (prescription needed) - Rapamycin Longevity News

Rapamycin: Rapamycin, mTOR, Autophagy & Treating mTOR Syndrome: Pelton, Ross: 9781607660156: Amazon.com: Books
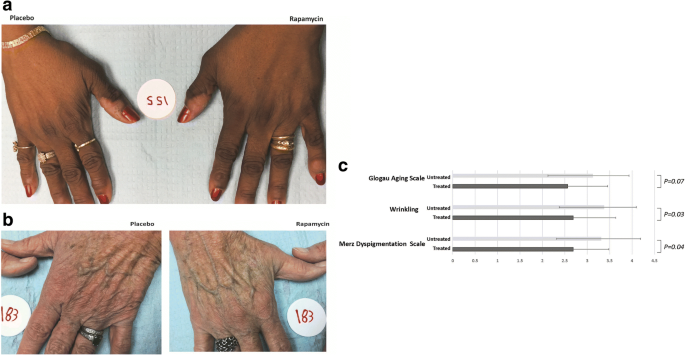
Topical rapamycin reduces markers of senescence and aging in human skin: an exploratory, prospective, randomized trial | GeroScience

Effects of the concurrent use of the 0.2% sirolimus gel and topical... | Download Scientific Diagram
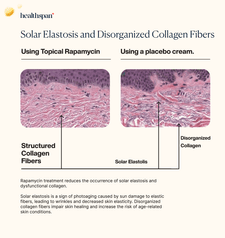
Healthspan Research Review | Inhibiting Skin Senescence with Topical Rapamycin (Sirolimus): A Novel Approach to Anti-Aging Skincare

Safety and Efficacy of the Sirolimus Gel for TSC Patients With Facial Skin Lesions in a Long-Term, Open-Label, Extension, Uncontrolled Clinical Trial | Dermatology and Therapy

Sirolimus-Coated Balloon in an All-Comer Population of Coronary Artery Disease Patients: The EASTBOURNE Prospective Registry | JACC: Cardiovascular Interventions
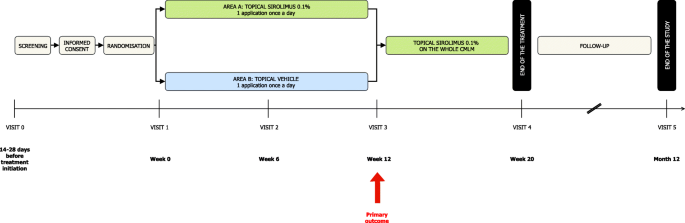
Topical sirolimus 0.1% for treating cutaneous microcystic lymphatic malformations in children and adults (TOPICAL): protocol for a multicenter phase 2, within-person, randomized, double-blind, vehicle-controlled clinical trial | Trials | Full Text

Biomedicines | Free Full-Text | Efficacy and Safety of Topical Mechanistic Target of Rapamycin Inhibitors for Facial Angiofibromas in Patients with Tuberous Sclerosis Complex: A Systematic Review and Network Meta-Analysis

Long‐term treatment of cutaneous manifestations of tuberous sclerosis complex with topical 1% sirolimus cream: A prospective study of 25 patients | Semantic Scholar

PDF) Bioprocess Engineering Aspects of Rapamycin (Sirolimus) Production: A Review on Past Achievements and Recent Perspectives